Viral Vector-Based Gene Therapy to Power Up Rare Disease Research
A pioneering shift in the research, diagnosis, and treatment of rare genetic diseases is on the horizon, powered by viral vector-based gene therapy. The one-stop development of gene therapy viral vectors, including the design and construction of suitable viral vectors as well as the production and purification of viral vectors, is unveiled in the quest to treat rare genetic disorders wherein traditional methods of treatment yield limited results.
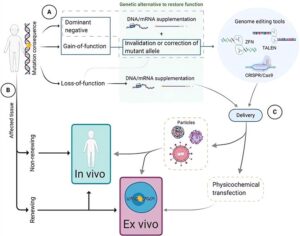
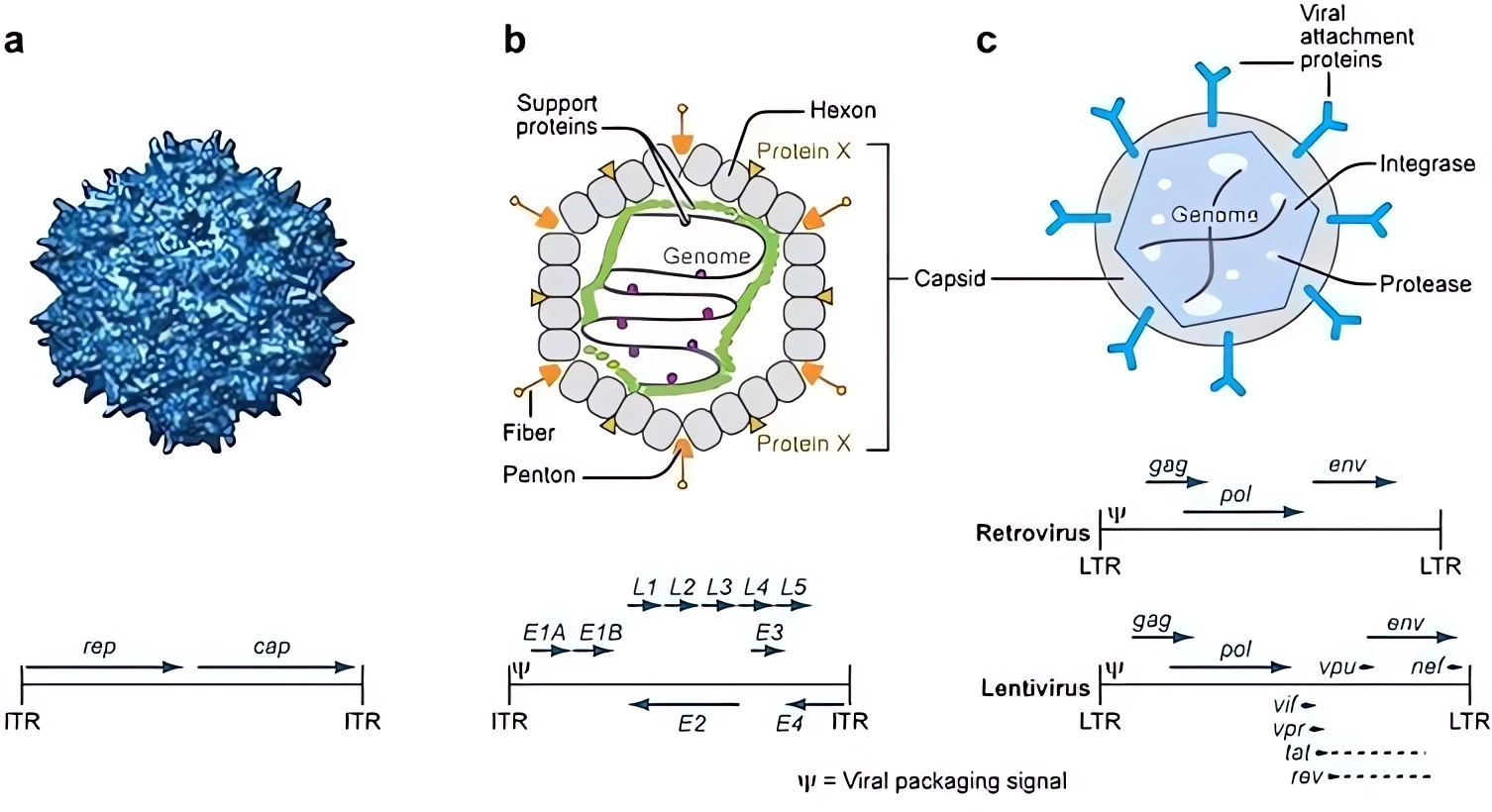
Gene therapies often necessitate the use of a vector to facilitate the delivery of the therapeutic gene-drug to the designated cell. Viral vectors are frequently studied due to their exceptional transduction efficacy and other impressive benefits. Clinically, gene therapy using viral vectors has been successful, with more than twelve viral gene therapy treatments approved to treat cancer, infectious diseases, and rare disorders. And the primary viral vectors employed in clinical in vivo gene therapy comprise adenovirus, adeno-associated virus (AAV), herpes simplex virus (HSV), retroviruses, and lentiviruses.
Gene therapy viral vectors can help to illuminate pathways in treating genetic disorders, demonstrating promise that will alter the paradigms of rare disease research. By leveraging the power of modified viruses, the healthy genes are delivered into a patient’s cells, replacing or correcting malfunctioning genes that cause disease. This treatment method not only addresses the symptoms but strikes at the very root of these disorders—the genetic mutations themselves.
“Gene therapy is the future of treating genetic diseases. With the development of advanced viral vector-based gene therapy, researchers are at the forefront of giving new hope to patients and their families, for whom treatment options have been historically limited,” says a senior scientist engaged in rare disease research. “We are excited by the clinical potential of viral vectors as a treatment platform for a multitude of genetic disorders.”
Motivated by more and more gene therapy approvals, comprehensive viral vector development is gearing up for the next stages of clinical evaluation, directed towards ensuring that this method is safe and effective in humans. Viral vector-based gene therapy gives researchers exceptional power not only to understand the intricacies of genetic diseases but, more importantly, to treat and potentially cure these diseases.